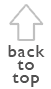Abstract 
Many
assessments of biodiversity are based on presence/absence data from
only a few taxa used as bioindicators, or at best a suite of
indicators from disparate taxonomic groups (for whom patterns of
diversity may not coincide). Community-based evaluations of
biodiversity can allow stronger comparisons between habitats or
indicate changes in population parameters by providing valuable
ecological information in addition to usual diversity/abundance
measures. Here we describe the use of communities of trap-nesting
Hymenoptera as bioindicators for ecological change. Additionally, we
give examples of data that can be obtained using exposure of
standardized trap nests in the target habitat. These data include
measurements of parasitism rates (decreasing with forest distance)
and foraging time (decreasing with resource availability). A case
example of the use of trap-nesting Hymenoptera is also provided as
part of a multidisciplinary investigation into the effects of land
use on biodiversity in mega-diverse regions of coastal Ecuador. This
investigation addresses the little known contribution of non-reserve
area to the preservation of biodiversity in the Ecuador.
In
particular it examines and compares biological diversity of different
land-use types (forest, agroforest, pasture or arable land such as
rice). Diversity assessments are made on plant, insect and bird
communities and the relationships between soil characteristics and
plant communities are also addressed. The utility of these taxa as
bioindicators is also examined. Furthermore, economic costs and
benefits associated with each land use system are estimated, with the
purpose to find financial instruments for biodiversity conservation.
Keywords: Land-use modeling, biological control, pollination, Hymenoptera,
agroforestry, conservation
Resumen 
Muchos estudios de
biodiversidad incluyen datos de presencia/ausencia de taxa
indicadores, o una serie de indicadores de grupos taxonómicos
independientes (de los cuales los patrones de biodiversidad no están
necesariamente relacionados). Evaluaciones de biodiversidad basados
en comunidades pueden proveer importante información ecológica
además de las usuales medidas de diversidad/abundancia, lo
cual permitiría una mejor comparación entre hábitats.
Aquí describimos el uso de comunidades de himenóptera
que anidan en "trampas-nido" como bioindicadores más
amplio. Adicionalmente se presentan ejemplos de datos que pueden ser
obtenidos usando esta técnica; estos datos incluyen medidas de
tiempo de forrajeo y tasas de parasitismo. Un ejemplo del uso de
"trampas-nido" para himenóptera es presentado como
parte de una investigación multidiciplinaria sobre los efectos
de los diferentes tipos de uso de suelo en la biodiversidad en
regiones megadiversas de la costa ecuatoriana. Esta investigación
trata sobre las dificultades asociadas con la preservación de
la biodiversidad en áreas no protegidas. En particular examina
y compara la diversidad biológica de diferentes sistemas de
uso de suelo (bosques naturales, sistemas agroforestales, pastizales
o cultivos de ciclo corto). Se evalúa la diversidad mediante
comunidades de plantas, insectos y aves, y se investiga la relación
entre las características del suelo y las comunidades
vegetales. Además se examina la utilidad de estos taxa como
bioindicadores. Finalmente, se estima los costos y beneficios
económicos asociados con cada sistema de uso de suelo con la
finalidad de establecer instrumentos financieros que contribuyan con
la conservación de la biodiversidad.
Palabras clave:
Ordenando el uso de tierra, control biológico, polinización,
Hymenoptera, agroforestería, conservación
Introduction 
The central problem surrounding direct measurement of biodiversity is the sheer number of species in most habitats, making indirect measurement techniques more heavily relied upon. Indirect measures generally incorporate presence/absence data from few taxa, such as lichens or aquatic invertebrates, as a basis for extrapolations to community-level diversity (e.g., Wolseley et al. 1994; Larsen et al. 1996). Several indicator taxa from different groups may even be used; however, these taxa may not show congruent patterns of diversity (Prendergast et al. 1993). This makes the selection itself of indicator taxa a partial determinant of the end diversity estimate. However, bioindicator groups can also be used in a broader sense to indicate changes in population parameters, ecological functions or community structure (e.g., Tscharntke et al.1998; Paoletti 1999; Davis et al. 2001). Insects make particularly useful bioindicators because they account for more than half of all species and their diversity allows for fine-scale resolution when differentiating between habitats.
Many surface-dwelling arthropods are used for bioindication because a) the taxa most often collected (such as carabid and staphylinid beetles and spiders) are polyphagous predators, and are therefore considered to be important for biological control, b) collections are made easily with pitfall traps, and c) catches are normally sufficiently large to allow statistical analyses (Duelli et al.1999). Pitfall sampling for ground-dwelling invertebrates may however lead to taxonomic difficulties and often-unmanageable sample sizes. While these problems can be partially overcome with simpler survey techniques (e.g., Andersen et al. 2002), such surveys yield ultimately information only on individual and species number, without any ecological context regarding species interactions. These ecological interactions are integral to the concept of biodiversity, and their exclusion reduces ecosystem diversity to a simple number, which does not contribute fully to the understanding of ecosystem services in multitrophic communities (Montoya et al.2003). For this reason Tscharntke et al. (1998) emphasize the importance of using community-based studies for evaluations of biodiversity. For example, marine crustacean, soil arthropod or trap-nesting Hymenoptera (Hymenoptera: Apidae, Eumenidae, Sphecidae and Pompilidae) communities make useful bioindication tools, due to their discreet area, trophic complexity and species diversity (Sanchez-Moyano & Garcia-Gomez 1998; Tscharntke et al. 1998; van Straalen 1998). In terms of ecosystem services (sensu Costanza et al. 1997) or functional agricultural biodiversity (Gurr et al. 2003), communities of trap-nesting bees and wasps are particularly salient, as their constituents are important pollinators of both crops and wild plants (Corbet et al. 1991; Klein et al. 2003). Anthropogenic declines in insect pollinators may have important consequences for plant biodiversity and stability of food crop yields (Allen-Wardell et al. 1998). Moreover, many trap-nesting species are also predators or parasitoids, thereby acting as biological control agents (e.g., Harris 1994). We will therefore discuss the use of trap-nesting Hymenoptera as bioindicators, and give examples of their use for biodiversity evaluation. We will also introduce a multidisciplinary project that examines the effects of land-use on soil- and biological diversity (especially trap-nesting Hymenoptera) and links them to their economic value.
Trap-nesting bees, wasps, and their enemies, examples of data
Methods 
Trap-nests suitable for occupation by Hymenoptera can be readily constructed, and make an easily manipulated, standardized community with which to compare habitats (Gathmann et al.1994; Tscharntke et al. 1998). A tin or PVC tube with a length of 22 cm and a diameter of approximately 15 cm forms the outer case of the nest. Internodes of reeds with varying diameter (2 - 20 mm) are inserted into this tube and provide the nesting sites for bees and wasps. Common reed Phragmites australis (Cav.) Trin. ex Steudel (Poaceae) or Japanese knotweed Polygonum cuspidatum Sieb. Et Zucc (Polygonaceae) are normally used, and they should be cut slightly shorter than the outer case (i.e. to approximately 20 cm) to allow some protection from rain. Trap-nests can be hung from trees in forest habitats or suspended from wooden posts in more open environments. Sticky glue (tanglefoot) is usually applied to the post or attachment point to deter ants. For more detailed methodology and statistical analyses see Klein et al. (2004).
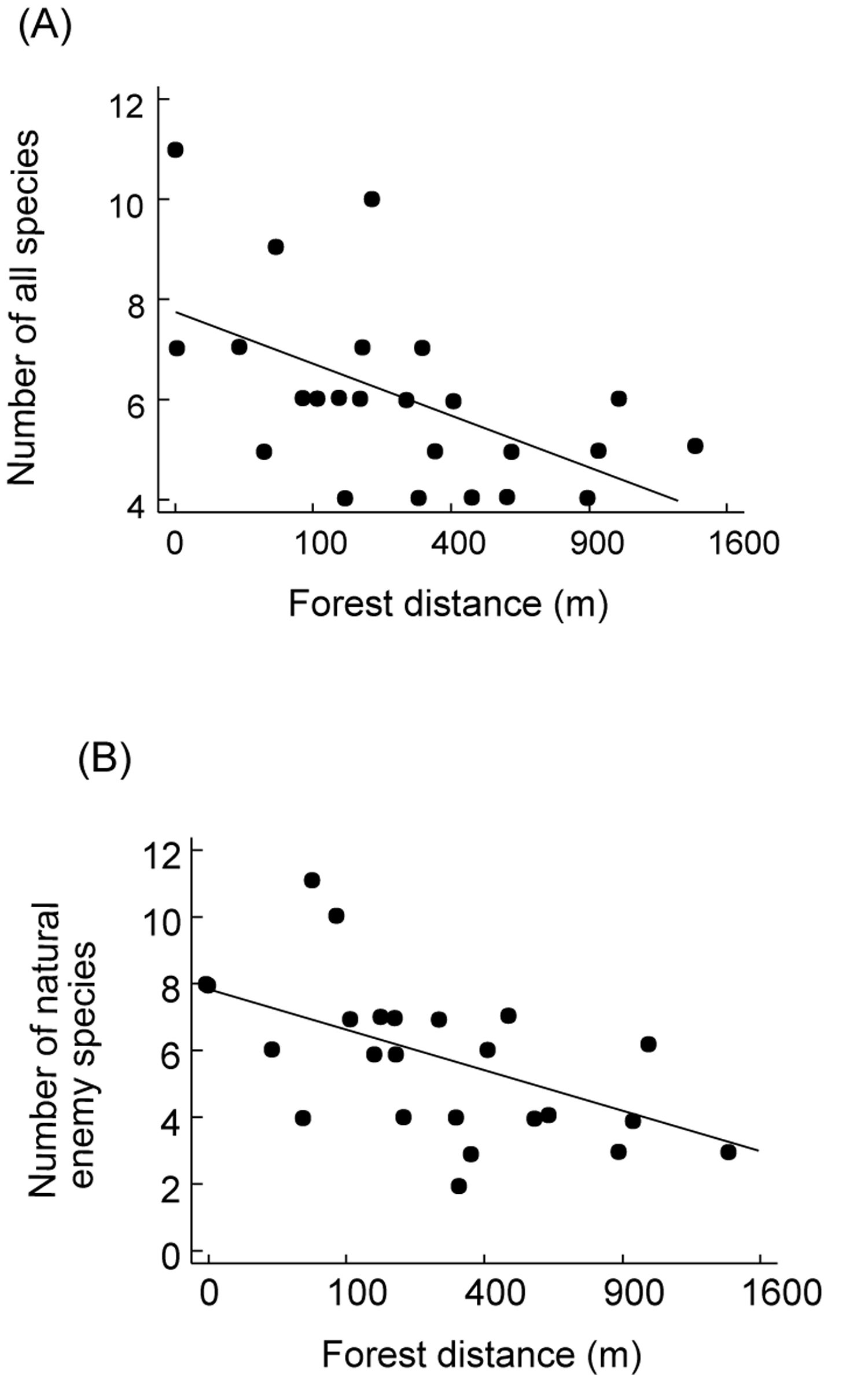
Figure 1. Effect of distance from nearest forest
fragment on (A) overall species richness per agroforestry system and
(B) number of parasitoid species per agroforestry system (taken from
Klein et al. Unpubl.).
Figura 1. Efecto de distancia de parcelas de bosque más cercanas de los sistemas agroforestales sobre (A) número de todas las especies por sistema agroforestal y (B) número de parasitoides por sistema agroforestal (tomado de Klein et al. Sinpubl.).
Figura 1. Efecto de distancia de parcelas de bosque más cercanas de los sistemas agroforestales sobre (A) número de todas las especies por sistema agroforestal y (B) número de parasitoides por sistema agroforestal (tomado de Klein et al. Sinpubl.).
Results and Discussion 
Trap-nests yield a suite of ecological information that can be used to compare habitats. As with other sampling techniques, they provide measures of overall abundance (Klein et al. 2002), and species richness ([[Figure 1A]]), as well as species richness of certain guilds, such as parasitoids (Figure 1B). These parameters can be used for habitat comparisons based on differences in successional stage (Gathmann et al. 1994), plant species richness (Tscharntke et al. 1998), land-use intensity (Klein et al. 2002) or habitat diversity (Steffan-Dewenter 2002). Moreover, trap-nests provide important and often-neglected information on ecological interactions such as the type of food provided to offspring (e.g., pollen, spiders, Lepidoptera larvae), rates of survival/parasitism (Figure 2), or foraging time (Figure 3). The latter is measured by observation of individual nests and recording of the time taken by females to acquire food and return to the nest. This is an important measure of habitat quality, as female bees in barren environments may require more time to locate food resources than females in environments where food resources are plentiful (Gathmann et al. 1994).
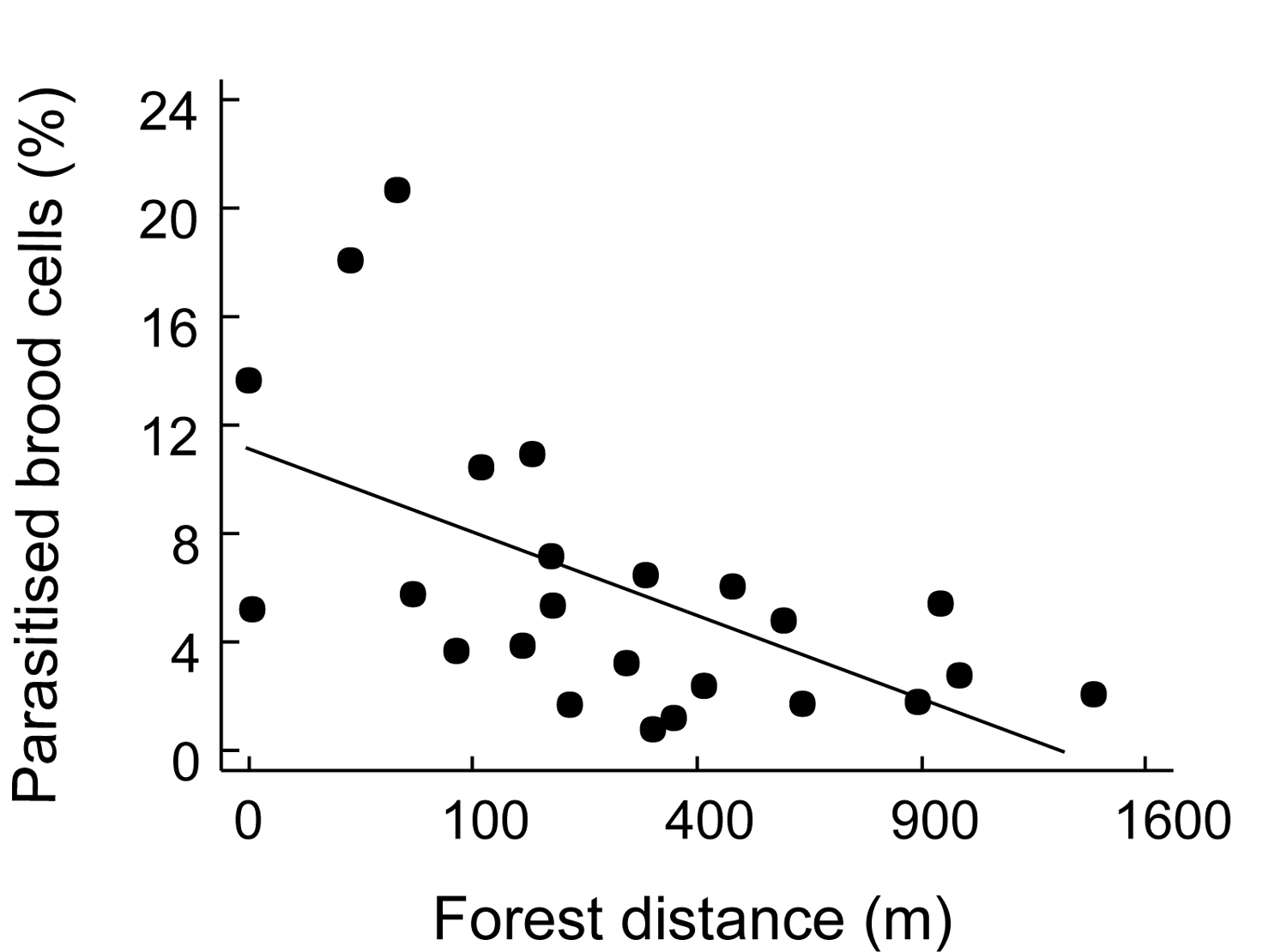
Figure 2. Effect of forest distance on the percentage
of trap-nesting Hymenoptera brood cells parasitized per agroforestry
system (taken from Klein et al. unpubl.).
Figura 2. Efecto de distancia de bosque sobre el porcentaje de crías parasitadas en trampas-nido de Himenóptera por sistema agroforestal (tomado de Klein et al. Sinpubl.).
Figura 2. Efecto de distancia de bosque sobre el porcentaje de crías parasitadas en trampas-nido de Himenóptera por sistema agroforestal (tomado de Klein et al. Sinpubl.).
Communities of trap-nesting bees and wasps are being currently used to compare different land-use types in Ecuador. This work is part of a multidisciplinary project that aims to assess the effects of land-use on biodiversity. We will briefly outline this project here.
Evaluation
of biological diversity of land-use systems in mega-diverse regions
of Coastal Ecuador, multidisciplinary project using the trap-nesting
technique
List of special symbols
ü - lower case u with umlaut. Pages 1, 12
ö - lower case o with umlaut. Page 1
Figure 3. Effect of blossom cover on foraging time of
Heriades (Michenerella) sp. aff. fulvescens (taken from Klein et al.
2004).
Figura 3. Efecto de cobertura floral sobre tiempo de búsqueda de alimento de Heriades (Michenerella) sp. aff. fulvescens (tomado de Klein et al. 2004).
Figura 3. Efecto de cobertura floral sobre tiempo de búsqueda de alimento de Heriades (Michenerella) sp. aff. fulvescens (tomado de Klein et al. 2004).
Rationale/components
Sustainable land use may produce a great variety of ecological goods and services (Altieri 1999). However, without payments for these services, environmentally-friendly land use may remain economically unattractive. Therefore, some kind of economic incentive may be necessary to encourage ecologically sustainable land use. Before incentives such as payment for ecosystem services can be initiated, however, information is needed regarding both the land management practices that are most beneficial for preservation of biodiversity, and the financial costs or benefits associated with these practices.
The current project lies at the interface of science and politics. We aim to evaluate species richness and ecosystem services in order to provide a scientific and operational basis for the implementation of payments for these services. Such payments, whether originating from the government or consumers, could potentially lift the economic burden of biodiversity preservation from landowners, allowing economically feasible conservation on private land.
The project is divided into four main components, one biological, one soil, one economic, and one land-use modeling. The aim of the diversity component is to determine the importance of differences in land-use types for the preservation of biodiversity and associated ecosystem function. Agroforests in particular may be important reservoirs for biodiversity (Perfecto et al. 1996; Moguel & Toledo 1999; Rice & Greenberg 2000; but see Rappole et al. 2003 for a caveat), and will therefore receive considerable attention. We will compare the effects of land-use, soil, and the interaction of these variables with plant communities and different insect taxa, and then examine the chain of effect from soil to plant communities to insects. Further investigations will examine bird diversity in natural forest and agroforestry systems, and relate this to food availability. The biodiversity of abandoned coffee agroforests will also be assessed and the effect of distance from natural forests on diversity will be measured to determine the importance of these areas as a source for species of conservation value.
The economic component will make cost/revenue comparisons and risk analyses for the different land-use types. The overall diversity (soil, plants, arthropods, birds) and economic components will then be integrated into a dynamic land-use model for the regions concerned.
The research is carried out in two provinces in Ecuador; Southern Manabí (a relatively dry region) and Southern Esmeraldas (a 'choco' region, characterized by very high rainfall and humidity). The land use types considered will be agroforestry (coffee in Manabí and cacao in Esmeraldas), pasture, and an arable crop (rice). In the diversity studies, natural forests in Esmeraldas and secondary forest fragments in Manabí (because natural rainforests no longer exist in this region) are used as a kind of positive control. The biodiversity of abandoned coffee agroforests will also be assessed and the effect of distance from natural forests on diversity will be measured to determine the importance of these areas as a source for species of conservation value.
The major components of the project are as follows:
Soil analyses
We will determine the effect of the interaction between soil type and land use management on diversity of chemical, physical and biological soil characteristics at the plot and landscape level and its relation with other ecosystem functions such as plant diversity, nutrient cycling and carbon sequestration.
Plant community analyses
We will use botanical surveys to map vegetation and determine the relationship between the diversity of plants and land-use type or combination of land use types. We will then relate plant diversity to diversity of trap-nesting bees, wasps, and their natural enemies as well as to bird diversity. Differences in plant community structure will also be related to soil characteristics at field and landscape level.
Bird diversity
Bird diversity in the different land use systems will be determined by means of the point count method in different seasons. Bird diversity in natural forest and agro-forestry systems will be related to food availability.
Insect community analyses
We will measure the effects of land-use types and management intensity on diversity and abundance of Hymenoptera. Measurements will be made using two methods, trap-nests and sweep-nets, to ensure reliability of results. Data from Manabí will be compared with that from Esmeraldas, so that the effect of climate/region on Hymenoptera diversity and abundance can be ascertained. Subsequently, we will determine the effects of differences in Hymenoptera diversity on ecosystem services such as coffee pollination and biological control of coffee pests.
We will also examine the effects of land-use on foraging behavior of trap-nesting Hymenoptera.
Economic
The economic component of the project focuses on comparisons for different land-uses and land management practices, as well as quantification of opportunity costs for biodiversity conservation. Farmers' land allocation problems are studied in detail considering that net revenues and risks are major determinants for land-use choice. Risk-analysis techniques include Montecarlo simulations and Stochastic Dominance. The biodiversity component of the overall research is integrated in this analysis by means of possible payments for environmental services, which leads to relevant policy implications.
Dynamic modeling
We will combine locally recorded ecological and economic information into a land-use model for the Manabi and Esmeraldas regions. This will allow regional-scale forecasts concerning land-use changes generated by financial incentives for providing eco-system services. Through scenario studies we identify potential trade-offs between biodiversity conservation, carbon sequestration and the production of agricultural and forestry products. Finally, it will be estimated how and where conservation measures can be implemented with the lowest opportunity costs. The model will be assigned from the local to the regional scale.
In conclusion, we plan to relate our detailed estimations of biodiversity to ecosystem services (such as biological control and pollination) and soil properties . The political value of our findings will depend on cost-benefit analyses and land-use modeling showing potential trade-offs between biodiversity conservation, carbon sequestration and productive crop management.
References 
Allen-Wardell, G.; P. Bernhardt; R. Bitner; A. Burquez; S. Buchmann; J. Cane & P.A. Cox et al. 1998. The potential consequences of pollinator declines on the conservation of biodiversity and stability of food crop yields. Conservation Biology 12: 8-17.
Altieri, M.A. 1999. The ecological role of biodiversity in agroecosystems. Agriculture, Ecosystems and Environment 74: 19-31.
Andersen, A.; B. Hoffmann; W. Müller & A. Griffiths 2002. Using ants as bioindicators in land management: simplifying assessment of ant community responses. Journal of Applied Ecology 39: 8-17.
Corbet, S.; L. Williams & J. Osborne 1991. bees and the pollination of crops and wild flowers in the European community. Bee World 72: 47-59.
Costanza, R.; R. d'Arge; R. de Groot; S. Farber; M. Grasso & B. Hannon et al. 1997. The value of the world's ecosystem services and natural capital. Nature 387: 253-260.
Davis, A; J. Holloway; H. Huijbregts; J. Krikken; A. Kirk-Spriggs & S. Sutton 2001. Dung beetles as indicators of change in the forests of northern Borneo. Journal of Applied Ecology 38: 593-616.
Duelli, P.; M. Obrist & D. Schmatz 1999. Biodiversity evaluation in agricultural landscapes: above-ground insects. Agriculture, Ecosystems and Environment 74: 33-64.
Gathmann, A.; H-J. Greiler & T. Tscharntke 1994. Trap-nesting bees and wasps colonizing set-aside fields: succession and body size, management by cutting and sowing. Oecologia 98: 8-14.
Gurr, G.; S. Wratten & J. Luna 2003. Multi-function agricultural biodiversity: pest management and other benefits. Basic and Applied Ecology 4: 107-116.
Harris, A. 1994. Ancistrocerus gazelle (Hymenoptera: Vespoidea: Eumenidae): a potentially useful biological control agent for leafrollers Plantortix octo, P. exessana, Ctenopseustis oblique, C. herana, and Epiphyas postvittana (Lepidoptera: Tortricidae) in New Zealand. New Zealand Journal of Crop and Horticultural Science 22: 235-238.
Klein, A-M.; I. Steffan-Dewenter; D. Buchori & T. Tscharntke 2002. Effects of land-use intensity in tropical agroforestry systems on coffee flower-visiting and trap-nesting bees and wasps. Conservation Biology 16: 1003-1014.
Klein, A-M.; I. Steffan-Dewenter; T. Tscharntke 2003. Pollination of Coffea canephora in relation to local and regional agroforestry management. Journal of Applied Ecology 40: 837-845.
Klein, A-M.; I. Steffan-Dewenter & T. Tscharntke 2004. Foraging trip duration and density of megachilid bees, eumenid wasps, and pompilid wasps in tropical agroforestry systems. Journal of Animal Ecology 73: 517-525.
Larsen, J.; H. Birks; G. Raddum & A. Fjiellheim1996. Quantitative relationships of invertebrates to pH in Norwegian river systems. Hydrobiologia 328: 57-74.
Moguel, P & VM Toledo 1999. Biodiversity conservation in traditional coffee systems of Mexico. Conservation Biology 13: 11-21.
Montoya, J.; M. Rodriguez & B. Hawkins 2003. Food web complexity and higher-level ecosystem services. Ecology Letters 6: 587-593.
Paoletti, M. 1999. Using bioindicators based on biodiversity to assess landscape sustainability. Agriculture, Ecosystems and Environment 74: 1-18.
Perfecto, I.; R. Rice; R. Greenberg & M. Van der Voort 1996. Shade coffee: a disappearing refuge for biodiversity. BioScience 46: 598-608.
Prendergast, J.; R. Quinn; J. Lawton; B. Eversham & D. Gibbons 1993. Rare species, the coincidence of diversity hotspots and conservation strategies. Nature 365: 335-337.
Rappole, J.; D. King & J. Vega Rivera 2003. Coffee and conservation. Conservation Biology 17: 334-336.
Rice, R. & R. Greenberg 2000. Cacao cultivation and the conservation of biological diversity. Ambio 29: 167-173.
Sanchez-Moyano, J. & J. Garcia-Gomez 1998. The arthropod community, especially Crustacea, as a bioindicator in Algeciras Bay (Southern Spain) based on spatial distribution. Journal of Coastal Research 14: 1119-1133.
Steffan-Dewenter, I. 2002. Landscape context affects trap-nesting bees, wasps, and their natural enemies. Ecological Entomology 27: 631-637.
Tscharntke, T.; A. Gathmann & I. Steffan-Dewenter 1998. Bioindication using trap-nesting bees and wasps and their natural enemies: community structure and interactions. Journal of Applied Ecology 35: 708-719.
Van Straalen, N. 1998. Evaluation of bioindicator systems derived from soil arthropod communities. Applied Soil Ecology 9: 429-437.
Wolseley, P.; C. Moncrieff & B. Aguirrehudson 1994. Lichens as indicators of environmental stability and change in the tropical forests of Thailand. Global Ecology and Biogeography Letters 4: 116-123.