Introduction

Mangrove
forests, dominated by estuarine trees serve as ecotones between land
and sea and elements from both are stratified horizontally and
vertically, between the forest canopy and subsurface soil (Rao &
Deshmukh, 1994). Mangrove has been defined as "any woody,
tropical halophyte that is an obligate inhabitant of 'mangal'
(wetland community) (Tomlinson ,1986). The word mangrove has
traditionally been used to describe either the total community or the
individual tree/ bushes, growing in the clayey, silty, inter-tidal
coastal zones, deltaic and estuarine coasts and backwaters/ sheltered
regions, in the tropical/subtropical belts of the world (Nayak &
Bahuguna, 2001). Mangroves can often survive non- saline habitats
(Cintron & Schaeffer-Novelli, 1983; Walsh, 1974). However,
according to Lugo (1980), a saline environment is required for stable
mangrove ecosystems. About 54-70 species (including hybrids) in 20-27
genera and 16-19 families fit comfortably into this broad category
(Tomlinson, 1986; Cronquist, 1981; Duke, 1992). Mangrove areas have
wide range of families, including ferns, grasses, sedges, palms and
legumes.Mangroves grow throughout the tropics wherever the average
monthly minimum temperature is 200 C (Chapman, 1976)and
are believed to be limited in their subtropical distribution by lack
of low temperature resistance (Dodd et al. 1995). Between 250
N and 250 S, mangroves colonize almost 75 % of the
coastline (Day et al., 1987) although they only represent 1 % (100000
km2) of the area of tropical forest are quite productive
(350 to 500 gram C m-2 yr-1) (Mann, 1982).
Mangroves may show strong, weak or no spatial zonation (Tomlinson,
1986; Ellison et. al., 2000), although the abundance of individual
species may follow the gradient of salinity (Helalsiddiqui, 1999).
Mangroves prefer a salinity range of 5- 30 parts per thousand.
The
ecological importance of these ecosystems for maintaining marine life
was stressed by Upadhyay et al. (2002); Fromard et al. (1998); Odum &
Heald (1975). Studies have demonstrated their role in supplying
organic material to coastal marine ecosystems (Odum & Heald,
1972; Lugo et al. 1980; Boto & Bunt, 1981; Rojas-Beltran, 1986;
Hutching & Saengar, 1987). Mangrove ecosystems are being studied
with more interest worldwide because of their economic importance in
support of commercial fisheries alone (Cintron et al. 1980). Uses and
values of mangroves are many and varied. For example, they provide
habitat as well as spawning and nursery ground for various marine
species (fish, shellfish, crustaceans etc), enrich the near-shore
environment, act as windbreakers and protects the shoreline from
storms, stabilize the shoreline, and decrease coastal erosion (Nayak
& Bahuguna, 2001).
Out of
4,87,100 ha. of mangrove wetlands in India, nearly 56. 7 % (2,75,800
ha.) is present along the east coast, 23. 5 % (1,14,700 ha.) along
the west coast, and the remaining 19.8 % (96,600 ha.) is found in the
Andaman and Nicobar islands (FSI,1999),. Mangroves in the densely
populated East Coast of India have been degraded for decades and are
still continuing to be degraded due to loss of biomass, species
composition simplification mainly due to overgrazing, fuel wood
extraction and conversions (Blasco & Aizpuru, 2002).
Mangroves are
spread over an area of 214 sq. km (FSI, 1999) in Orissa. The
assessment has indicated an increase of 20 sq. Km (FSI, 1997, 1999)
in Bhadrak and Kendrapara districts. Although the overall assessment
shows an increase, several areas have shown marked decrease in
quality and quantity of the vegetation cover. Causes for degradation
of mangroves in Orissa are, shoreline changes, settlements,
conversion for agriculture and aqua culture (Upadhyay et al. 2002).
Recent researches carried out on biosystematics of mangrove
phanerogams (Dodd et. al. 1995; Duke, 1995; Tomlinson, 1986); on
biogeography (Saengar, 1996); ecology (Snedaker, 1995) and
distribution (Spalding et al. 1997) can be considered as being of
direct interest to the knowledge of the mangroves of India (Blasco &
Aizpuru, 1997). Taxonomical works on mangroves have been done by
Banerjee et al. (1989), Banerjee (1984, 1987); Banerjee & Rao
(1990); Choudhury (1984, 1990); Choudhury et al. (1991, 1995);
Majumdar & Banerjee (1985); Mishra & Panigrahi (1987).The
present paper highlights ecological structures of mangroves ecosystem
of Orissa coast based on phytosociological studies.
The Study Site
The state of
Orissa has a geographical area of 155707 sq. km with an actual forest
cover of 47107 sq. km. (30.3 %). Area under Mangrove forests is 195
sq. km which comes to 0.125 % of geographical area and 0.414 % of
actual forest cover (Daniels & Acharjyo, 1997). The study site
located at 200 4'- 200 8' N
Latitude and 860 45'- 870 5' E
Longitude, in the north-eastern coastal plain of Kendrapara district
in Orissa is in Bhitarkanika sanctuary. Total area of sanctuary is
672 Sq. km of which mangrove forests constitute 130 sq. km. This area
receives water from three rivers, known to be rich in species
diversity and trees are dense and tall like those of Sunderbans
(Selvam, 2003). Four forest blocks in the Bhitarkanika wildlife
sanctuary were selected for carrying out vegetation survey. The area
of Bhitarkanika forest block is 1712 ha., Dangmal 636 ha., Kakranasi
310 ha., and Thakurdia 272 ha. (Chadha & Kar, 1999). Bhitarkanika
and Dangmal Bocks constitute the core area. These sites experience
tide of semi diurnal type. The mean sea level in the region is about
1. 66 meters. The Bhitarkanika sanctuary is bounded by river Dhamra
in the north, the river Hansua to the west and Bay of Bengal on the
eastern and southern sides. The sanctuary encompasses 35 km sea coast
known as 'Gahirmatha Coast' from Dhamra mouth to Barunei,
the mouth of river Hansua. The area has about 200 km. of water body
inside the sanctuaryand falls in the deltaic region of the river
Brahmani, Baitarani, and their tributaries. The estuarine rivers-
Brahmani, Baitarani, Kharasrota, Dhamra, Pathasala, Maipura, Hansua,
and Hansina during their course flow into the Bay of Bengal are
further criss crossed by numerous creeks, channels, and nallahs, thus
providing the peculiar ecological niche for the growth, development
of rich and varied mangrove life forms, both flora and fauna along
with their associates. There are many villages within the sanctuary
as well as surrounding it. The population in these villages has been
growing very fast. Part of the population rise is because of the
heavy influx of refugees from East Bengal and West Bengal and
habitations are reported to have been started by clearing mangrove
forests. A total of 81 villages are adjacent to the mangrove forests.
The population increase is attributed as one of the reasons for
decreasing mangrove of the area.
Climate
The region
comes under the tropical monsoon climate with three pronounced
seasons: winter (October to January), summer (February to May) and
rainy (June to September). The maximum temperature is recorded in the
month of April and May and the minimum temperature in winter during
the month of January. The relative humidity ranges from 70% to 84%
through out the year. Wind speed from March to June is over 20 km.
per hour, and the predominant wind direction is from south and
south-west. Rainfall is around 1642.34 cm per annum and maximum
rainfall is received between June and October. The most important
weather phenomenon is the prevalence of tropical cyclones. The mean
track of the cyclone passes over this region (Singh & Panda,
1999). Rainfall conditions decide the sequence of mangrove
distribution in the different zones in the tidal region. A successive
tidal flood inundates the land surface and the subsequent exposure of
the soil substratum evaporates the water. This result in thick salt
crust on the soil surface and these salt crusts inhibit or limit the
regeneration and growth of the mangroves. Frequent rainwater flushing
helps in washing off the surface and leaching down the salt particles
and makes the land suitable for growth of mangroves. Tidal amplitude
in the Baunsagada River ranges from 1.5 to 2.5 meters in summer
months to 3 to 5 meters during monsoon months. In the Bhitarkanika
River, and especially in creeks such as Khola (which receives tidal
water from both ends) tidal amplitude reaches 3- 4 meters in summer
months to 5-6 meters during rainy season.
Soils and Geology
The soil
sediments are divided into two categories, indicating recent or
sub-recent forms named as 'newer alluvium' and
Pleistocene forms named as 'older alluvium' (GSI, 1974).
The recent sediments are represented by sand, silt, and clay with
assorted boulders and pebbles. These are dark and loosely compacted
with high moisture content. The Pleistocene deposits comprise of
clay, sand, silt, and 'kankar', with locally cemented
pebbles and gravels. These are reddish brown due to high degree of
oxidation (Banerjee & Rao, 1990).
Methods

Phytosociological
Analysis in four forest blocks was carried out by quadrat method
following Misra (1968), Kershaw (1973), Cintron and Schaefer-Novelli
(1984) and Snedaker & Snedaker (1984). Thirty quadrats of 10m X
10 m size were laid out at each site. Each site was divided into 6
segments of 1 km each along tidal line from the riverbank. A line
transect was laid towards landward side from the water line. In each
segment, 6 quadrats of 10 m X 10 m size were laid at 0, 50, 100, 150,
200 and 250 meter interval towards the land ward side for
phytosociological analysis. 120 quadrats were laid in four forest
blocks to study forest structure (trees). On the basis of data
obtained from quadrat samples, the structural parameters like
frequency, abundance, density, basal area, and IVI were calculated
(Tables 1a-d).
Results and Discussion

Vegetation Analysis
The
Bhitarkanika forest block contains highest number of tree species
followed by Dangmal, Kakranasi and Thakurdia blocks. Bhitarkanika and
Dangmal are part of core area of the Bhitarkanika wildlife sanctuary.
Availability of fresh water through Bhitarkanika (Maipura river) and
Brahmani rivers and saline water from sea in core area help wide
range of niches for different species to occur and, thus, species
diversity is the highest. Table 1 provides details on structural
parameters of vegetation of study sites. H. fomes and E.
agallocha exhibited greater density, frequency and IVI values
across all sites. The species with lower density and IVI are
different from one site to the other. All the species show contagious
distribution. A/F ratio range in Dangmal block is proportionately
less wide compared to other blocks. According to Odum (1971)
contagious distribution is commonest in nature, random distribution
is found only in very uniform environment and regular distribution
occurs where severe competition exists between individuals.
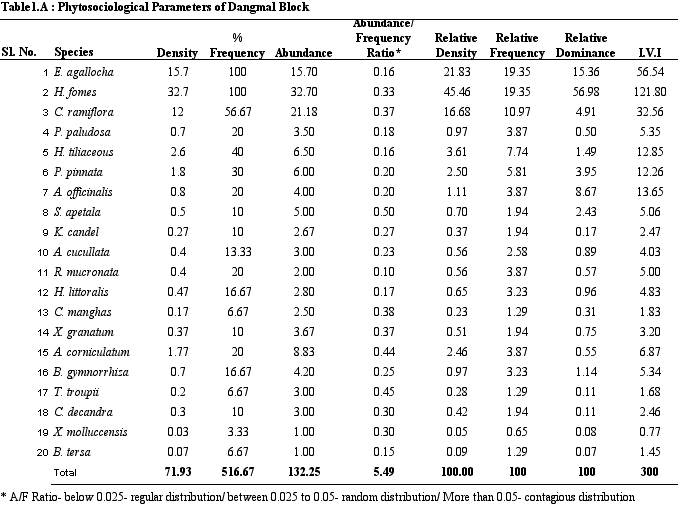
Table 1A.
Phytosociological parameters of Dangmal Block
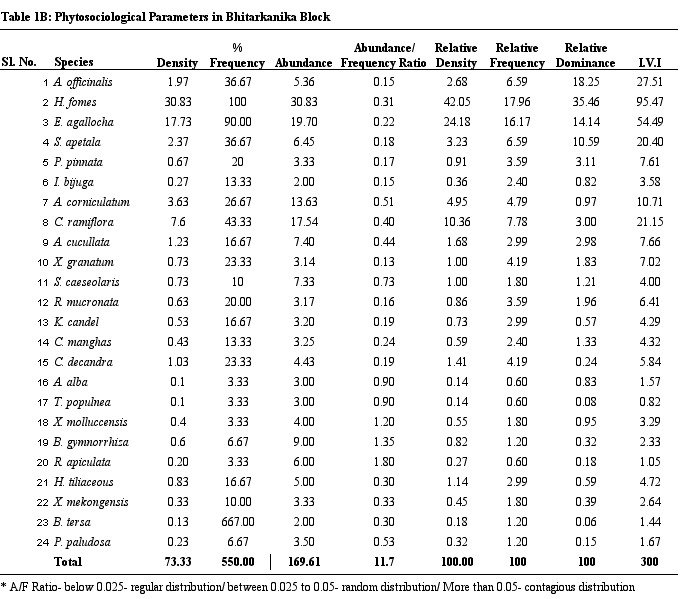
Table 1B.
Phytosociological parameters of Phitarkanika Block
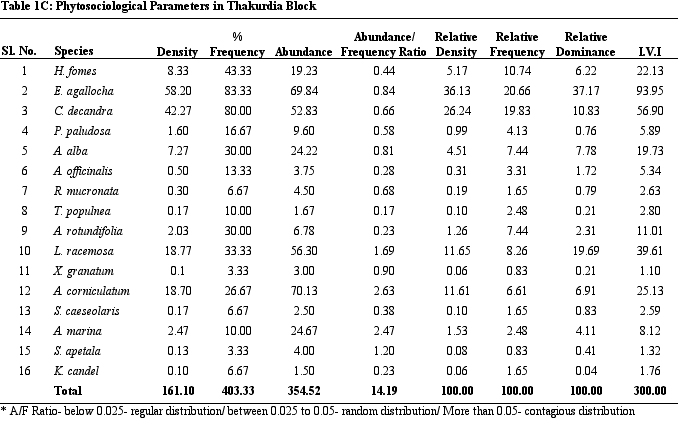
Table 1C.
Phytosociological parameters of Thakurdia Block
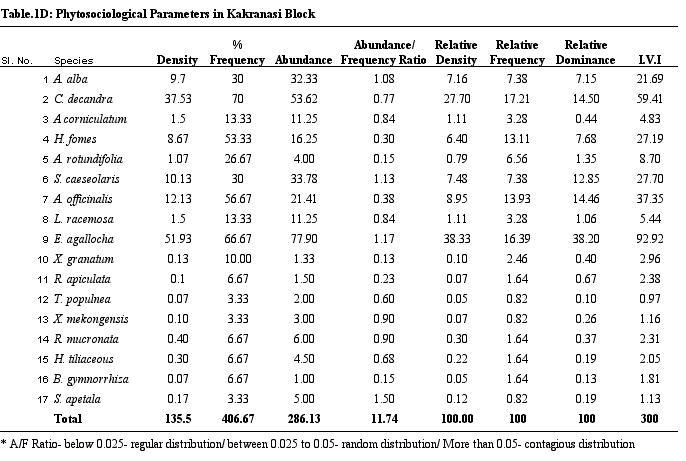
Table 1D.
Phytosociological parameters of Kakranasi Block
From
list of species encountered through quadrat surveys of trees and
seedlings (Table 2), it is observed that the family Rhizophoraceae
and Meliaceae represented maximum number of species followed by
Avicenniaceae. Bhitarkanika is the most species rich site with 24
species and Thakurdia has the lowest species number with 16 species
(Table 3). Bhitarkanika has the highest mean species value per
quadrat (5.56 species per quadrat). The average value for all the
forest blocks is 4.69 species per quadrat. Ellison (2002) established
a correlation between latitude and longitude and species richness and
observed that the species richness is higher (> 30 to 55 species)
between 0 and 200 N lat and at 70 and 1350 E
long. Species richness is highest in the Indo West Pacific and
declines relatively smoothly from 1000 E which is the
longitude of peak species richness (Ellison et al. 1999).
Table 2. Mangrove and associated species in the study
area
|
Sl No
|
Species encountered
through quadrat survey
|
Species encountered
through seedling survey
|
Other species
encountered during survey
|
|
Dicotyledons
|
|
|
|
1
|
Acanthaceae
|
|
|
|
|
Acanthus ilicifolius
|
|
|
2
|
Aizoaceae
|
|
|
|
|
Sesuvium
portulacastrum
|
|
|
3
|
Apocynaceae
|
|
|
|
Cerbera manghas
|
Cerbera manghas
|
|
|
4
|
Avicenniaceae
|
|
|
|
Avicennia alba
|
|
|
|
Avicennia
officinalis
|
Avicennia
officinalis
|
|
|
Avicennia marina
|
|
|
|
5
|
Caesalpiniaceae
|
|
|
|
|
Caesalpinia crista
|
|
|
Cynometra ramiflora
|
Cynometra ramiflora
|
|
|
Intsia bijuga
|
Intsia bijuga
|
|
|
6
|
Chenopodiaceae
|
|
|
|
|
|
Salicornia brachiata
|
|
|
|
S. maritima
|
|
7
|
Combretaceae
|
|
|
|
Lumnitzera racemosa
|
|
|
|
8
|
Euphorbiaceae
|
|
|
|
Excoecaria agallocha
|
Excoecaria agallocha
|
|
|
9
|
Malvaceae
|
|
|
|
Thespesia populnea
|
Thespesia populnea
|
|
|
Hibiscus tiliaceous
|
Hibiscus tiliaceous
|
|
|
10
|
Meliaceae
|
|
|
|
Amoora cucullata
|
|
|
|
Xylocarpus granatum
|
Xylocarpus granatum
|
|
|
Xylocarpus
mekongensis
|
Xylocarpus
mekongensis
|
|
|
Xylocarpus
molluccensis
|
Xylocarpus
molluccensis
|
|
|
11
|
Myrsinaceae
|
|
|
|
Aegiceras
corniculatum
|
Aegiceras
corniculatum
|
|
|
12
|
Papilonaceae
|
|
|
|
|
Dalbergia spinosa
|
|
|
Pongamia pinnata
|
Pongamia pinnata
|
|
|
13
|
Peripocaceae
|
|
|
|
|
|
Finlaysonia obovata
|
|
14
|
Plumbaginaceae
|
|
|
|
Aegialitis
rotundifolia
|
|
|
|
15
|
Rhizophoraceae
|
|
|
|
Bruguiera
gymnorrhiza
|
Bruguiera
gymnorrhiza
|
|
|
Ceriops decandra
|
Ceriops decandra
|
|
|
Kandelia candel
|
Kandelia candel
|
|
|
Rhizophora apiculata
|
Rhizophora apiculata
|
|
|
16
|
Rutaceae
|
|
|
|
|
|
Merope angulata
|
|
17
|
Salvadoraceae
|
|
|
|
|
|
Salvadora persica
|
|
18
|
Sonneratiaceae
|
|
|
|
Sonneratia apetala
|
Sonneratia apetala
|
|
|
Sonneratia
caeseolaris
|
Sonneratia
caeseolaris
|
|
|
19
|
Sterculiaceae
|
|
|
|
Heritiera fomes
|
Heritiera fomes
|
|
|
Heritiera littoralis
|
Heritiera littoralis
|
|
|
20
|
Tamaricaceae
|
|
|
|
Tamarix troupii
|
Tamarix troupii
|
|
|
21
|
Tiliaceae
|
|
|
|
Brownlowia tersa
|
Brownlowia tersa
|
|
|
22
|
Verbenaceae
|
|
|
|
|
|
Clerodendrum inerme
|
|
Monocotyledons
|
|
|
|
23
|
Arecaceae
|
|
|
|
Phoenix paludosa
|
Phoenix paludosa
|
|
|
24
|
Flagellariaceae
|
|
|
|
|
|
Flagellaria indica
|
|
25
|
Poaceae
|
|
|
|
|
Myriostachya
wighitiana
|
|
|
|
Porteresia coarctata
|
|
|
26
|
Polypodiaceae
|
|
|
|
(fern)
|
Acrostichum aurum
|
|
A total of 22
families of Dicotyledons and 4 families of Monocotyledons were
represented across all sites in Bhitarkanika Mangrove ecosystem. A
total of 43 species of mangrove and associated plants belonging to 32
genera were recorded from 26 families of Angiosperms. The flora is
extremely diverse in the estuarine regions of Bhitarkanika, (Banerjee
& Rao 1985, 1990).Abundance of phanerogams is presumably higher
than that of the Sunderbans Gangetic delta. Though the factors
influencing biodiversity and floristic richness in each deltaic
region is not fully understood (Duke et al. 1998), the assumption is
that the propagules originated in the Sunderbans are water buoyant
and dispersed to the nearest deltaic area which is the mouth of the
Mahanadi river. This could explain the high degrees of relationship
between the flora of the Gangetic and the Mahanadi deltas (Blasco &
Aizpuru, 2002). H. fomes, Sonneratia. griffithii and Aegialitis
rotundifolia Roxb. are endemic to the coastal part of South Asia
(Blasco et al. 2001) and later two species are not recorded during
the study.
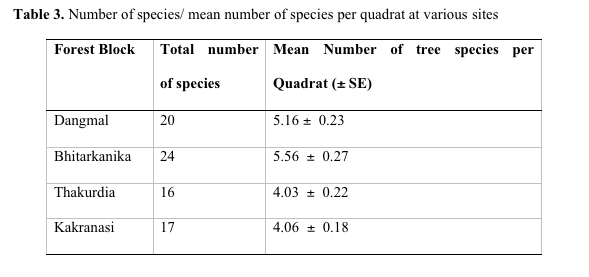
Table 3. Number of
species / mean number of species at various quardat sites
Raunkiaer's Frequency Class Distribution
Raunkiaer's
Law of Frequency (in graphical form referred to as Raunkiaer's
J shaped distribution curves) was studied (Raunkiaer, 1934). The law
(also known as the law of homogeneity) was expressed as A>
B > C ≤≥D
E, wherein, A to E are
frequency classes suggested by Raunkiaer's from 0 to 100.
According to Kershaw (1973), "the increase in class E reflects
the theoretical infinite range of density and contrasts with the more
strictly defined limits for classes A, B, C, and D. This E class has
a density range greatly exceeding frequency classes A to D.
Accordingly many more species fall into this class, despite the
general tendency for 'common' species to be relatively
few in number in a community"(Fig.1 ).
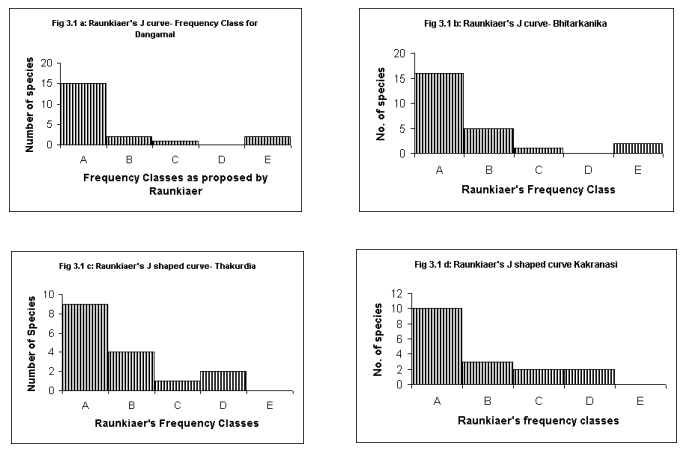
Raunkiear's
Frequency classes for the study sites
Species Diversity
The
species diversity depends upon adaptation of species and increases
with stability of community (Singh et al, 1994). Species diversity
was 0.72, 0.82, 0.75, 0.73, respectively, in Dangmal, Bhitarkanika,
Thakurdia, and Kakranasi blocks. The above data indicate that
Bhitarkanika site is highly diverse and Dangmal the least. The
Concentration of Dominance was 0.28, 0.25, 0.23, 0.24, respectively,
in Dangmal, Bhitarkanika, Thakurdia, and Kakranasi indicating the
dominance is more pronounced in Dangmal block (Table 4). The Dangmal
and Kakranasi blocks exhibited least similarity in species
composition (59.46 %) with each other followed by Thakurdia and
Bhitarkanika (65 %), Thakurdia and Kakranasi (78.79 %), and
Bhitarkanika and Dangmal blocks (86.36%) (Table 5). The latter two
sites are adjacent to each other and thereby there is a great deal of
species mix. In the Eastern hemisphere number of mangrove
species reported by Tomlinson (1986) and Duke (1992) are 58 compared
to only 12 in Western hemisphere. High mangrove diversity in South
East Asian region is because it has been the center of origin of
mangrove speciation. There is presence of adjacent diverse
terrestrial flora which has enabled diversity to increase and
prevented extinctions (Ricklefs & Latham, 1993). Duke et al.
(1998) found the Indo-Malaysia region with most mangrove species
number with 48 species.
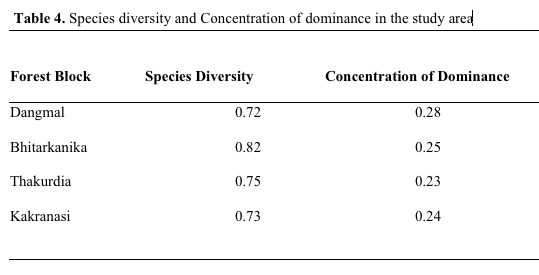
Table 4. Species
diversity and concentration of dominance in the study area
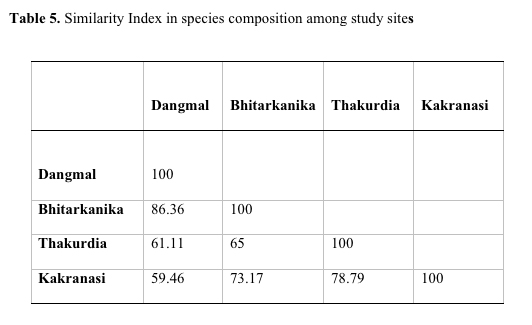
Table 5.Similarity
index in species composition between study sites
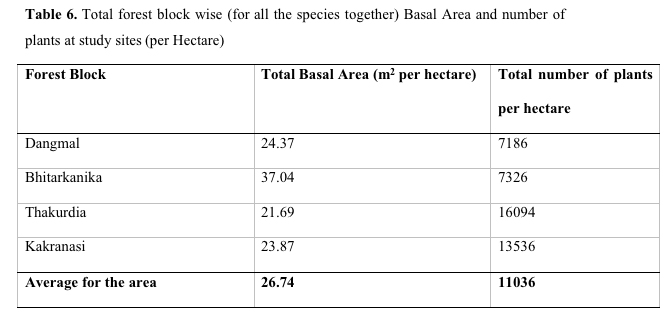
Table 6. Total Basal
Area and number of species per study site
The
species diversity is higher in the India mangrove ecosystems compared
to that of Latin America and Africa. Large physical forces in
tidewater, salinity level, and lack of stable substratum are some of
the natural factors that affect the species diversity (Pathway et al.
2002).Studies on the changes in the species composition for
Bhitarkanika are not available like other mangrove areas on the east
coast i.e., Sunderbans, Pichavaram and Muthupet, Guava, and Andaman &
Nicobar Islands (Kannupandi & Kannan, 1998; Caratini et al.,
1973; Mathuda, 1959; Azariah et al. 1992). H. fomes is known to
require low soil and water salinities. When the salinity increases,
the species becomes stunted, rare, and ultimately disappears. It is
known to be 'top dying' (trees shedding their leaves due
to stress and could be dying) in parts of Bangladesh(Siddiqi, 1998)
and Sunderban because of the increase in dry season demand for
freshwater, damming of rivers and apparent downstream effects of
increase in soil salinities (Blasco et al. 2001). Therefore, this
species is a leading dominant in the mangroves of Bhitarkanika, and
thereby confirms to the availability of good ecological conditions
that harbours it well. However, caution has to be exercised to see
that the preconditions that are now suitable continue to be so. In
the mangrove areas of Myanmar H. fomes was available in plenty
between the mouth of Mayu and Lamu city about 50 years back and has
been completely depleted due to high salinity stress (Blasco, et al.
2001). Others have also reported about die back of Heritiera fomes
due to adverse increase in soil salinity (Christensen & Snedaker,
1984; Chaffey et al. 1985).
Several authors have worked on
phytosociological parameters of Tropical Mangroves. In French Guiana
forests Fromard et al. (1998) observed that in mature coastal and
adult riverine mangrove sites Avicennia exhibited the highest
value of IVI (144 - 181) followed by Rhizophora species.
These mangrove types are more frequent in Guiana and are homogenous
and dominated by A. germinatus. The mangroves on sea
fronts generally have high basal area (24.6 - 33.6 m2
ha-1). The riverine mangrove ecosystems are more
diversified and mixed type and richer in species having tree density.
However, the species, H. fomes and E. agallocha exhibited
dominance with high value of IVI followed by A. officinalis in
the mangroves of Orissa coast at Bhitarkanika.
References

Azariah, J, V. Selvam & S. Gunasekaran. 1992. Impact of past
management practices on the present status of the Muthupet mangrove
ecosystem. Hydrobiologia 247: 253- 259.
Banerjee, L.K. 1984. Vegetation of Bhitarkanika sanctuary in Cuttack,
Orissa. India, Journal of Economic & Taxonomic Botany 5:
1065- 1079.
Banerjee, L.K. & T.A. Rao. 1985. Mangals of Mahanadi delta,
Cuttack district, Orissa state, India, in: Marine Plants, paper
presented at the All India Symposium on Marine Plants, Goa.
Banerjee, L.K., A.R.K. Sastry & M.P.Nayar. 1989. Mangroves in
India-identification manual. Botanical Survey of India, 113
pages.
Banerjee, L.K. & T.A. Rao. 1990. Mangroves
of Orissa Coast and their ecology. Bishen Singh Mahendra Pal
Singh, Dehra Dun, India, 118 pages.
Banerjee, L.K., 1987. Ecological studies on the
mangals of Mahanadi estuarine delta, Orissa. Tropical Ecology
82: 117- 125.
Blasco, F. & M. Aizpuru. 1997. Classification and evolution of
the mangroves of India. Tropical Ecology 38: 357- 374.
Blasco, F, M. Aizpuru & C. Gers. 2001. Depletion of the mangroves
of continental Asia. Wetlands Ecology and Management 9: 245-
256.
Blasco, F & M. Aizpuru. 2002. Mangroves along the coastal stretch
of Bay of Bengal: present status. Indian Journal of Marine Science
31: 9- 20.
Boto, K.G. & J.S. Bunt. 1981. Tidal export of particulate organic
matter from a southern Australian mangrove system. Estuarine
Coastal Shelf Sci. pp- 247- 255.
Caratini, C., F. Blasco & G. Thankaimoni. 1973. Pollen Spores
15: 281- 292.
Chadha, S. & C.S. Kar. 1999. Bhitarkanika: Myth and Reality.
Natraj Publishers, Dehradun, 368
Chaffey, D.R., F.R. Miller & J.H. Sandom. 1985. A forest
inventory of the Sunderbans, Bangladesh. Main Report, ODA,
Project Report, 140.
Chapman, V.J. 1976. Mangrove Vegetation. J. Cramer, Vaduz,
Liechtenstein.
Choudhury, B P. 1984. A glimpse into the vegetation of Bhitarkanika
wildlife sanctuary in the state of Orissa. India. Bot. Reptr.
3: 121- 124.
Choudhury, B.P. 1990. Bhitarkanika: mangrove swamps. Journal of
Environmental Science 3: 1- 16.
Choudhury, B.P., A.K. Biswal & H.N. Subudhi. 1991. Mangroves of
Orissa and aspects of their conservation. Rheedea 1: 62- 67
Choudhury, B.P., A.K. Biswal & S.P. Rath. 1995. Biodiversity in
the Bhitarkanika wildlife sanctuary in the state of Orissa. In: (R.C.
Mohanty (ed.). Environment: Change and management . New
Delhi, pp- 53- 60.
Christensen, B. & S.C. Snedaker. 1984. Integrated development
of the Sunderbans: ecological aspects of the Sunderbans. FAO:
Field Document No 3, FO: TCP/BGD/2309 (Mf), Rome.
Cintron, G.Y. & Y. Schaeffer-Novelli. 1983. Introduction a la
ecologia del manglar, Montevideo, UNESCO, 99 pages.
Cintron, G. & Y.S. Novelli. 1984. Methods for studying mangrove
structure. In: Samuel C. Snedaker & J.G. Snedaker- (eds.). The
mangrove ecosystem: research methods . UNESCO, 251 pages.
Cronquist, S. 1981. An integrated system of classification of
flowering plants. Columbia University Press, New York.
Day, J.W., W. Conner, F. Ley-Lou, R.H. Day & A.M. Navarro. 1987.
The productivity and composition of mangrove forests, Laguna de
Terminos, Mexico. Aquatic Botany 27: 267- 2844.
Dodd, R.S., F. Fromard, Z.A. Rafii & F. Blasco. 1995.
Biodiversity among West African Rhizophora, Foliar Wax Chemistry.
Biochemical Systematics and Ecology 23: 859- 868.
Duke, N.C.. 1992. Mangrove floristics and biogeography. In: A.I.
Robertson & D.M. Alongi (eds). Tropical Mangrove Ecosystems.
American Geophysical Union, Washington DC, pp- 63- 100.
Duke, N.C. 1995. Genetic diversity, distributional barriers, and
rafting continents- more thoughts on the evolution of mangroves.
Hydrobiologia 295: 167- 181.
Duke, N.C., M.C. Ball & J.C. Ellison. 1998. Factors influencing
biodiversity and distributional gradients in mangroves. Global
Ecol. Biogeogr. 7: 27- 47.
Ellison, A.M., E.J. Farnsworth & R.E. Merkt. 1999. Origins of
mangrove ecosystems and the mangrove biodiversity anomaly. Global
Ecol Biogeogr. 8: 95- 115.
Ellison, A.M., B.B. Mukherjee & A. Karim. 2000. Testing patterns
of zonation in mangroves: scale dependence and environmental
correlates in the Sunderbans of Bangladesh. Journal of Ecology
88: 813- 824.
Ellison, A.M. 2002. Macroecology of mangroves: large scale patterns
and processes in tropical coastal forests. Trees
16: 181- 194.
Forest Survey of India. 1999. State of Forest Report- 1999.
Dehra Dun
Fromard, F., H. Puig, E. Mougin, G. Marty, J.L. Betoulle & L.
Cadamuro. 1998. Structure, above- ground biomass and dynamics of
mangrove ecosystems: new data from French Guiana, Oecologia,
115: 39- 53.
GSI. 1974. Geological Survey of India Miscellaneous publication
no. 30.
Helalsiddiqui, A.S.M. 1999. Status of the major mangrove species in
the Sunderbans of Bangladesh. Indian Journal of Forestry22 :
197- 202.
Hutchings, P. & P. Saengar. 1987. Ecology of mangroves.
University of Queensland Press, St. Lucia.
Kannupandi, T. & R. Kannan. 1998. Anthology of Indian
Mangroves. ENVIS Center, Annamalai University, Chidambaram, pp-
25- 29.
Kershaw, K.A. 1973. Quantitative and Dynamic Plant ecology (second
edition), Edward Arnold, London.
Lugo, Ariel E. 1980. Mangrove ecosystems: successional or steady
state? Biotropica 12: 65- 72.
Lugo, A.E., R.R. Twilley & C. Patterson-Zucca. 1980. The role
of black mangrove forests in the productivity of coastal ecosystems
in south Florida. U. S. Environment Protection Agency,
Corvallis, Oregon.
Majumdar, N.C. & L.K. Banerjee. 1985. A new
species of Heritiera
(Sterculiaceae) from Orissa.Bulletin of Botanical Survey of India
27: 150- 151.
Mann, K.H. 1982. Ecology of coastal waters. University of
California Press, Berkeley.
Mathuda, G.S. 1959. Proceedings of the mangrove symposium.
Ministry of Food and Agriculture, Kolkata.
Mishra, S.C. & G. Panigrahi. 1987. Studies on the mangrove flora
of Orissa with particular reference to Rhizophoraceae. R. Br. J.
Econ. Tax. Bot.. 11: 121- 132.
Misra, R. 1968. Ecology Work Book. Oxford and IBH Publishing
Co./ New Delhi.
Nayak, S. & A. Bahuguna. 2001. Application of remote sensing data
to monitor mangroves and other coastal vegetation of India. Indian
Journal of Marine Sciences 30: 195- 213.
Odum, E.P. 1971. Fundamentals of ecology. W. B. Saunders,
Philadelphia.
Odum, W. & E.J. Heald. 1972. Trophic analysis of an estuarine
mangrove community. Bull. Marine Sci. 22: 671- 738.
Odum, W.E. & E.J. Heald, 1975. Mangrove forests and aquatic
productivity In: A.D. Hassler (Ed.). Coupling of Land and Water
Systems . Ecol. Stud., University of Virginia, Charlottesville,
10, pp- 129- 136.
Phillips, E.A. 1959. Methods of vegetation survey. Holt,
Rienhart & Winston Inc., New York.
Rao, A.N. & S. Deshmukh. 1994. In: Deshmukh, S.V. & V. Balaji
(Eds.). Conservation of mangrove forest genetic resources: a
training manual. ITTI-CRSARD project, M.S. Swaminathan Research
Foundation, Chennai, India.
Ricklefs, R.E. & R.E. Latham. 1993. Global patterns of diversity
in mangrove floras, in: R.E. Ricklefs & D. Schluter (Eds.).
Species Diversity in Ecological Communities. University of
Chicago Press, Chicago, pp- 215- 229.
Rojas-Beltran, R. 1986. Role de la mangrove comm. Nourricerie de
crustaces et de poissons en Guyane. In: Sepanguy-Sep-Anrit (Ed.). Le
Littoral Guyanais, fragilite de l'environment, ler Congres
Sepanguy Nature Guyanaise, Cayenne, pp- 97- 110.
Saengar, P. 1996. Mangrove flora: Distribution of species and habitat
descriptions. In: D.I. Walker, F.E. Wells, & J.R. Hawley (Eds.).
Marine biological survey of the Eastern Kimberley, Western
Australia. The University of W.A., W.A. Museum and the Museum of
Art Gallery of the Northern Territory, Perth, pp- 39- 53.
Siddiqi, N.A. 1998. Enrichment planting in the mangrove of
Sunderbans- Arcview, Bangladesh Journal of Forest Science
27: 103- 113.
Singh, D.K. & G.K. Panda. 1999. Bhitarkanika and its environs-
a geographical appraisal, in: Bhitarkanika- the wonderland of Orissa.
Nature and Wildlife conservation society of Orissa, Bhubaneswar,
India, pp- 10- 18.
Snedaker, S.C. & J.G. Snedaker,.1984. The mangrove ecosystem:
Research methods. UNESCO, Paris.
Spalding, M, F. Blasco & C.D. Field (Eds.). 1997. World
Mangrove Atlas. Japan: The International Society for Mangrove
Ecosystems. Cambridge Samara Pub. Co., Cambridge.
Tomlinson, P.B. 1986. The botany of mangroves. Cambridge
Tropical Biology Series, Cambridge University Press, 413 pages.
Upadhyay, V.P., R. Ranjan & J.S. Singh. 2002. Human mangrove
conflicts-the way out, Current Science 83: 1328- 1336.
Walsh, G.E. 1974. Mangroves: a review. In: R.J. Peinold & W.H.
Queen (Eds.). Ecology of Halophytes. Academic Press, New York,
USA, pp- 51- 174.
4